Professionals
At Keya Medical, we believe that medical imaging AI technology can benefit patients, healthcare providers, and payers. By focusing on these healthcare market segments, we offer potential investors an opportunity to drive the commercial development of medical imaging AI products that are expected to evolve to meet the needs of aging populations in the world’s largest economies.

Healthcare Delivery
Keya Medical is committed to developing medical imaging AI products in collaboration with healthcare providers. The solutions we develop in collaboration with clinical partners to improve the speed and accuracy of diagnosis, reduce unnecessary invasive testing, and enable physicians to deliver more personalized treatment to patients. Since 2016, our product development team has worked in collaboration with over 200 hospitals around the world to bring solutions to clinical use that solve real challenges.
Keya Medical is actively recruiting collaboration partners to help bring DEEPVESSEL FFR and DEEPVESSEL CARS to clinical use in the United States.
Collaboration opportunities
Collaborative Research
Keya Medical scientists work with clinicians to understand workflow and end-user needs to inform the development of new technology. De-identified image data from healthcare providers is used to train and validate deep learning models.
Clinical Effectiveness Studies
In collaboration with health systems, we work to perform clinical effectiveness studies to include in submissions to the FDA and other regulatory agencies.
Usability Studies
We work with medical and usability research professionals to conduct formal studies to ensure that our solutions are intuitive and safe for clinical use.

Payers
Keya Medical is continuing to develop new solutions to improve the health of populations with cardiovascular health issues.
The adoption of CT-derived Fractional Flow Reserve technology is an example of how healthcare payers are essential to the translation of major breakthroughs in patient care from research to clinical practice. The US Center for Medicaid and Medicare began reimbursing for its use in 2018, and this procedure is now recognized and paid for by many commercial insurance companies in the US and by the UK National Health Service. This is critically important because, in some markets, CT Angiograms compete with other diagnostic tests, such as Stress Echocardiograms, in clinical use even though an increasing body of evidence suggests that patients whose care is guided by CTA and CT derived FFR have better long-term outcomes.
We invite payers to continue the conversation with our team on how Keya Medical’s solutions can create savings.

Investors
Keya Medical’s funding has come from investors, including:
- IDG Capital
- GGV Capital
- Kunlun Internet Smart Fund
- Alwin Capital
- Growth FOF
R&D Product Roadmap
Our R&D scientists, developers, and designers are working to bring new solutions to healthcare that will improve the speed and accuracy of diagnosis, enabling physicians to deliver more personalized treatment to patients.
The following solutions are included in Keya Medical’s product development pipeline:
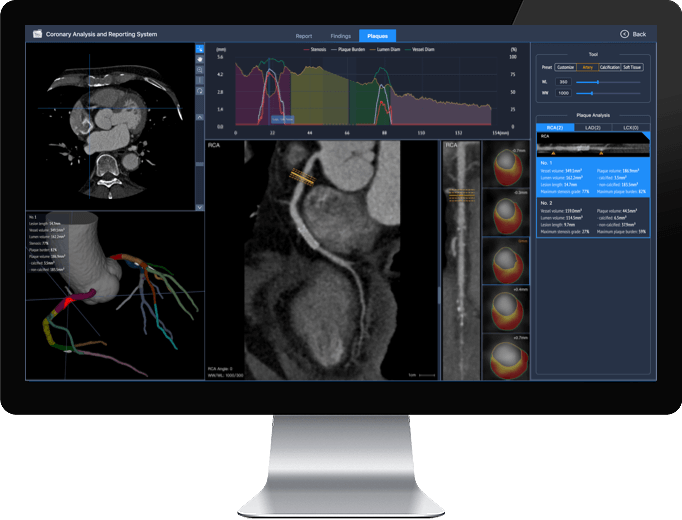
Coronary Analysis and Reporting System (CARS)
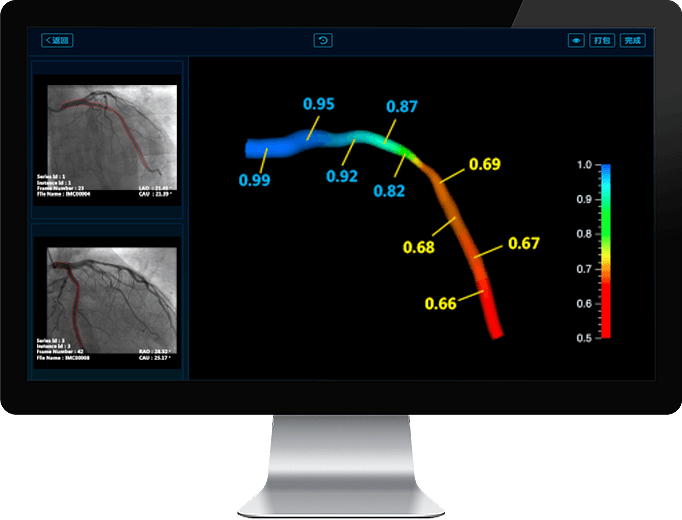
Adenosine and Wire-Free Artery Functional Assessment
Recent Funding
Keya Medical Raises $46M in Series D Round
Keya Medical has raised $46 million in Series D funding. This funding round was led by new and existing investors, including CICC Capital, Shanghai Artificial Intelligence Investment Fund, Qianhai Gaozu Asset Management Fund, and IN CAPITAL.
Keya Medical Closes $30 Million Series C Funding Round Led by IDG Capital
Keya Medical announced that it has completed a $30 Million Series C Funding Round led by IDG Capital, with participation from investors Alwin Capital, Source Code Capital, and Tasly Group.
Keya Medical Raises Over $20 Million in Series B+ Financing Round
Keya Medical announced on Aug. 13 that it has raised over $20 million in Series B+ funding.

Products
DeepVessel FFR
CuraRad-ICH
Solutions
All Solutions
Diagnostic Solutions
Treatment Solutions
Solution Suites