DEEPVESSEL FFR CT-Recent Peer-Reviewed Publications
Following is a sampling of recent peer-reviewed publications and more studies with DEEPVESSEL FFR CT are in progress.
Keya Medical presents its CT FFR analysis at SCCT2024.
Keya Medical showcases its deep-learning-based CT FFR analysis at SCCT2024. Contact us for more information or to schedule a personalized demonstration of our deep-learning-based CT FFR solution.
Join us at SCCT2024
Connect with the Keya Medical team at Booth 114, SCCT2024 Washington, D.C. or book a private meeting.
Kanbai Tech adds CT-FFR to its AI medical imaging platform.
Kanbai Tech furthers the implementation of Keya Medical’s CT-FFR physiological assessment technology in Spain.
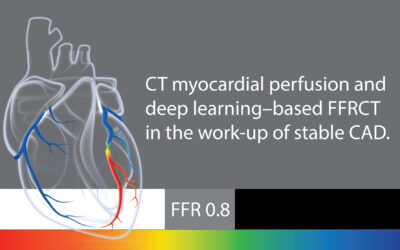
The Stepwise CCTA + FFRCT + CT-MPI Strategy: A Promising Approach for Evaluating Stenosis with Gray Zone FFRCT Values
2024 study to validate a stepwise strategy using FFRCT for intermediate stenosis on CCTA and CT-MPI for vessels with gray zone FFRCT values.
Keya Medical Joins Bayer’s Calantic AI Solution for Radiology
Keya Medical showcases its AI CT-FFR analysis through Bayer’s Calantic Digital Solutions platform at ECR2024.
CT-FFR: Enhancing Selectivity and Precision in Cardiac Patient Care
Selective use of CT-FFR successfully identified patients who would benefit most from invasive procedures, reducing unnecessary interventions.
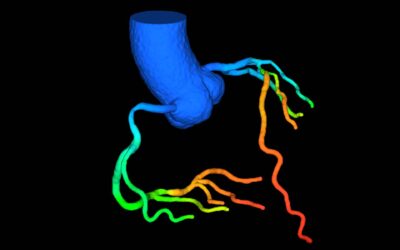
Advancements in AI-based CT-FFR Analysis with DEEPVESSEL FFR: A Comprehensive Research Overview
Keya Medical’s AI CT-FFR Analysis: focus on its research and publications journey since DEEPVESSEL FFR FDA-clearance.
Keya Medical at 2024 ECR Annual Congress-Next Generation Radiology.
Take advantage of the opportunity to participate in a personalized one-on-one discussion with Keya Medical and explore a product demonstration.
DEEPVESSEL FFR Achieves Global Milestone with Four Certifications
On December 27, 2023, Keya Medical's CT FFR product DEEPVESSEL FFR (DVFFR) passed the certification of the Health Sciences Authority (HSA) of Singapore and was officially launched in Singapore! This is a breakthrough for Keya Medical’s artificial intelligence medical...
Keya Medical Presents FDA-cleared CT FFR product at SCCT Global 2024 | Budapest
Visit Keya Medical’s booth at SCCT Global 2024 in Budapest, January 16-19.
Scaling your Cardiovascular CT Service and Implementing CT-FFR
Discover the power of automated CT-FFR integration in enhancing your cardiovascular CT services.