
Can FFR-CT Reduce Unnecessary Testing in Moderate CAD?
A recent study published in REC: Interventional Cardiology (2026) explores whether adding FFR-CT (DEEPVESSEL FFR) to standard CCTA can reduce testing in moderate CAD.

A recent study published in REC: Interventional Cardiology (2026) explores whether adding FFR-CT (DEEPVESSEL FFR) to standard CCTA can reduce testing in moderate CAD.

Keya Medical’s advanced AI solutions and the application of Deep Learning CT-derived Fractional Flow Reserve analysis in non-invasive cardiac diagnostics. Visit us at SCCT 2025, Booth 14.

Visit Keya Medical’s booth at SCCT Global 2024 in Budapest, January 16-19.

In the dynamic landscape of medical innovation, SCCT 2023 served as an exceptional platform for exchanging cutting-edge insights and breakthroughs in cardiovascular technology.

Keya Medical US celebrates the achievements of the team in China and their commitment to the commercialization of DEEPVESSEL FFR.
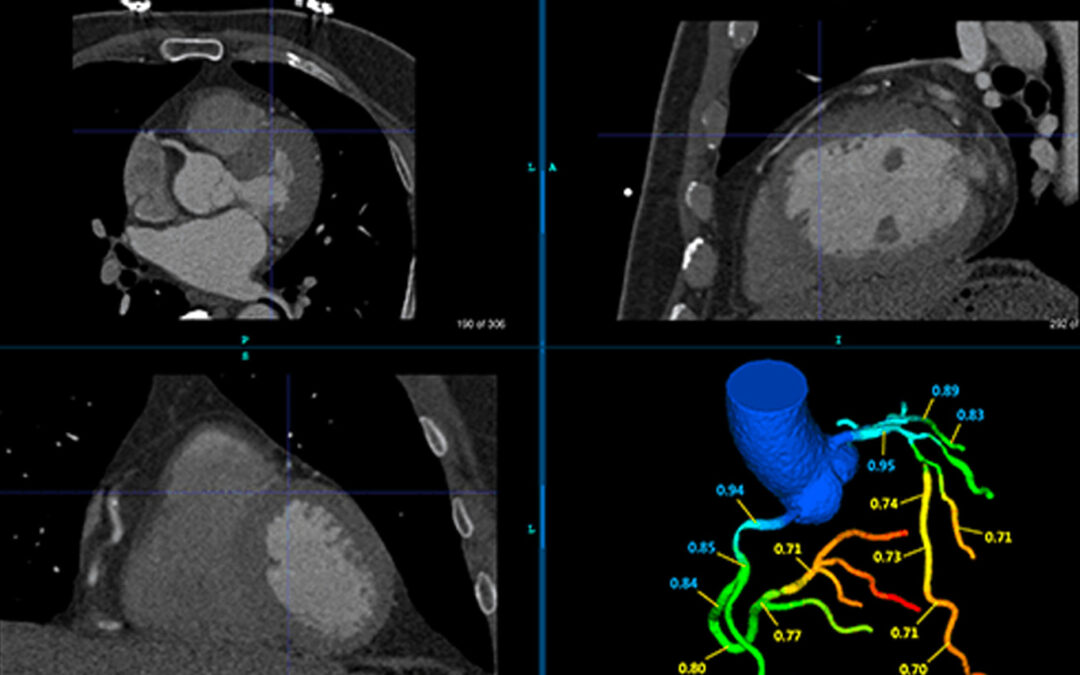
Seattle, WA– April 1, 2022– Keya Medical announced that DEEPVESSEL FFR (DVFFR), has been cleared by U.S. Food and Drug Administration (FDA).
Recent Comments