
by KeyaMedical | Nov 12, 2020 | All News, Investors, Press
Keya Medical announced that it has completed a $30 Million Series C Funding Round led by IDG Capital, with participation from investors Alwin Capital, Source Code Capital, and Tasly Group.

by KeyaMedical | Aug 23, 2020 | Investors, Press
Keya Medical announced on Aug. 13 that it has raised over $20 million in Series B+ funding.

by KeyaMedical | Feb 19, 2020 | Investors, Press
Keya Medical announced today that it has received a strategic investment from Kunlun Wanwei Group, IDG Capital, Shanghai Guofang Parent Fund, and Yahui Capital.
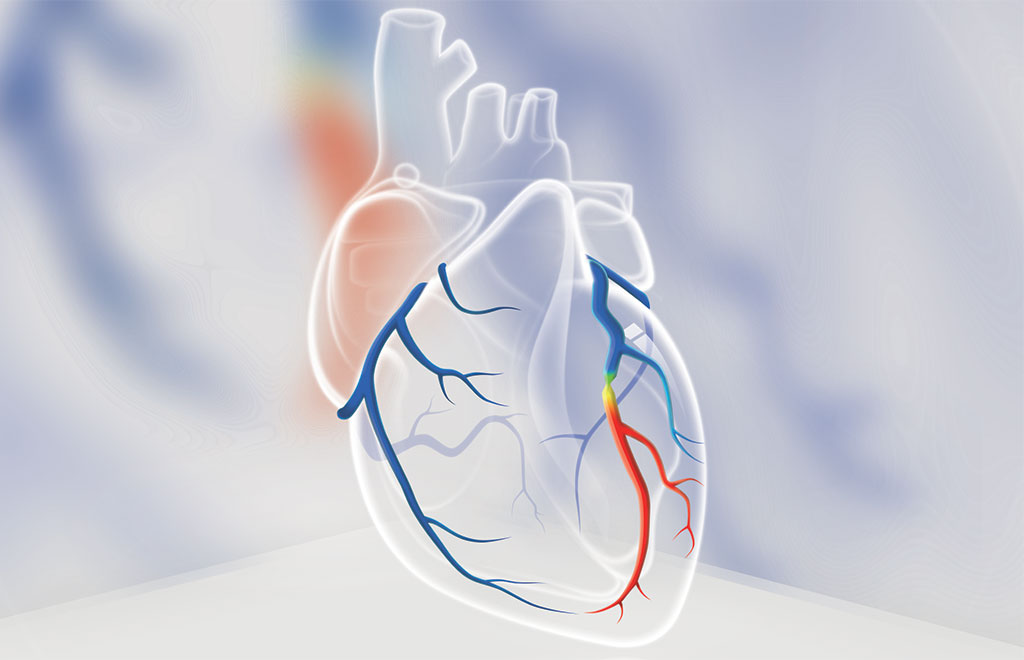
by KeyaMedical | Jan 22, 2020 | Investors, Press
On January 15, 2020, the National Medical Products Administration in China issued the registration certificate approving DeepVessel FFR for clinical use.
by KeyaMedical | Sep 21, 2018 | Press
Keya Medical announced on September 21, 2018 that DEEPVESSEL FFR received ISO 13485:2016 certification for Medical Device and Quality Managements Systems as well as its CE Mark Certification after a series of on-site audits and evaluations by its notified body, DEKRA.





Recent Comments