Keya Medical’s CT FFR analysis DEEPVESSEL® is now available in the USA.
Deep-learning-based, non-invasive, accurate, and accessible FFR CT analysis DEEPVESSEL FFR is now available in the USA.

Deep-learning-based, non-invasive, accurate, and accessible FFR CT analysis DEEPVESSEL FFR is now available in the USA.
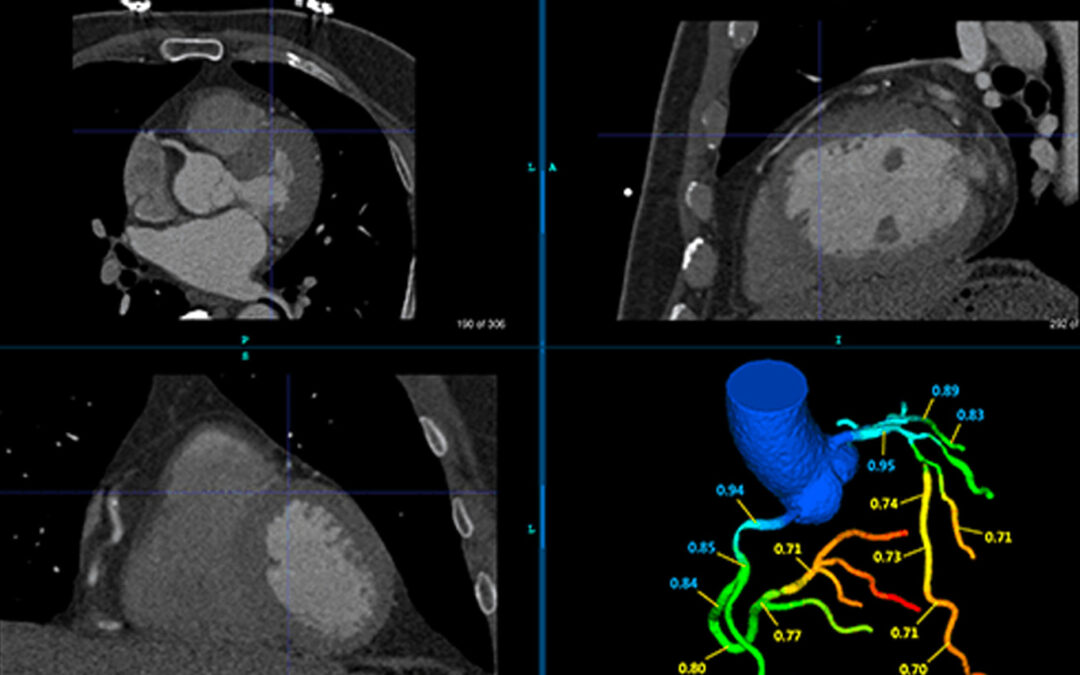
Seattle, WA– April 1, 2022– Keya Medical announced that DEEPVESSEL FFR (DVFFR), has been cleared by U.S. Food and Drug Administration (FDA).

Keya Medical showcased its deep learning-based diagnostic and treatment solutions at the World Artificial Intelligence Conference 2021 (WAIC 2021) held in Shanghai.

Keya Medical was recognized on the Top 100 Future Medical Companies List at the Fifth Future Healthcare VB100 Conference.
Keya Medical has received regulatory approval in China for its DeepVessel 3D Visualization Processing Software. The AI-based software medical device is designed for visualization reading of multiphase CT images and 3D models.

Keya Medical, a leading developer of medical AI technology, has received the Most Growth Enterprise Award granted by iiMedia Research, a third-party data mining and analysis company headquartered in China.
Recent Comments